Colchicine
Anti-cancer compound. Microtubule assembly inhibitor. Depolymerizes microtubules and limits microtubule formation (inactivates spindle fibre formation). Inhibits mitosis during cell division at metaphase by inhibiting spindle formation. Anti-inflammatory compound. Suppresses monosodium urate crystal-induced NLRP3/NALP3 inflammasome-driven caspase-1 activation, IL-1beta processing and release, and L-selectin expression on neutrophils at micromolar concentrations. Blocks the release of a crystal-derived chemotactic factor from neutrophil lysosomes, blocks neutrophil adhesion to, and inhibits monosodium urate crystal-induced production of superoxide anions from neutrophils at nanomolar concentrations. Drug used in treatment of gout, familial Mediterranean fever, pericarditis and Behect’s disease. Investigated for its anti-cancer activity. It has a narrow therapeutic index with no clear-cut distinction between nontoxic, toxic and lethal doses, causing substantial confusion among clinicians. Apoptosis inducer in a variety of normal and tumor cell lines. Inhibitor of autophagosome-lysosome fusion. Inhibits acetylated a-tubulin mediated dynein dependent transport of mitochondria and subsequent apposition of ASC on mitochondria to NLRP3 on the endoplasmic reticulum in vitro and in vivo.
| Catalog Number | AG-CN2-0048-G001 |
| Alternative Name(s) | NSC 757; EINECS 200-598-5 |
| Research Area | Apoptosis, Autophagy, Cancer, Cell Death, Inflammasomes, Inflammation, Natural Products |
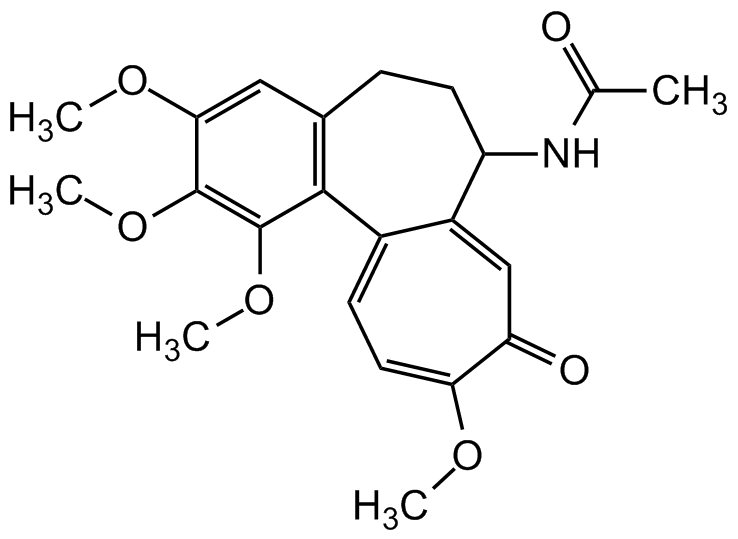
| Molecular Formula | C22H25NO6 |
| CAS# | 64-86-8 |
| Purity | >98% |
| Inchi | InChI=1S/C22H25NO6/c1-12(24)23-16-8-6-13-10-19(27-3)21(28-4)22(29-5)20(13)14-7-9-18(26-2)17(25)11-15(14)16/h7,9-11,16H,6,8H2,1-5H3,(H,23,24) |
| Inchi Key | IAKHMKGGTNLKSZ-UHFFFAOYSA-N |
| SMILES | COC1=CC2=C(C(OC)=C1OC)C1=CC=C(OC)C(=O)C=C1C(CC2)NC(C)=O |
| Size | 1 g |
| Supplier Page | http://www.adipogen.com/ag-cn2-0048/colchicine.html |

