TargetMol
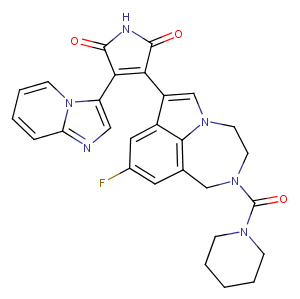
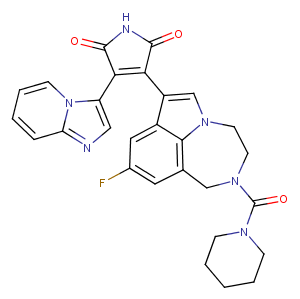
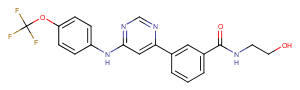
LY2090314, an effective GSK-3α/β inhibitor (IC50: 1.5 nM/0.9 nM), may improve the efficacy of platinum-based chemotherapy regimens. LY2090314 has been used in trials studying the treatment of Leukemia, Advanced Cancer, and Pancreatic Cancer.
More Information
Supplier Page
TargetMol
LY2090314, an effective GSK-3α/β inhibitor (IC50: 1.5 nM/0.9 nM), may improve the efficacy of platinum-based chemotherapy regimens. LY2090314 has been used in trials studying the treatment of Leukemia, Advanced Cancer, and Pancreatic Cancer.
More Information
Supplier Page
TargetMol
LY2090314, an effective GSK-3α/β inhibitor (IC50: 1.5 nM/0.9 nM), may improve the efficacy of platinum-based chemotherapy regimens. LY2090314 has been used in trials studying the treatment of Leukemia, Advanced Cancer, and Pancreatic Cancer.
More Information
Supplier Page
TargetMol
Carfilzomib is an irreversible proteasome inhibitor and antineoplastic agent that is used in treatment of refractory multiple myeloma.
More Information
Supplier Page
GNF-5
100 mg
| 99.78%
TargetMol
GNF-5 is a specific non-ATP competitive inhibitor of Bcr-Abl (IC50: 0.22±0.1 uM, Wild-type Abl). It is an analog of GNF-2 with improved pharmacokinetic properties.
More Information
Supplier Page
GNF-5
10 mg
| 99.78%
TargetMol
GNF-5 is a specific non-ATP competitive inhibitor of Bcr-Abl (IC50: 0.22±0.1 uM, Wild-type Abl). It is an analog of GNF-2 with improved pharmacokinetic properties.
More Information
Supplier Page
TargetMol
Ataluren
50 mg
| Purity Not Available
TargetMol
Ataluren is a novel, orally administered drug that targets nonsense mutations. Ataluren is approved for use by the European Medicines Agency to treat Duchenne Muscular Dystrophy in patients aged 5 years and older who are able to walk.
More Information
Supplier Page
Ataluren
200 mg
| Purity Not Available
TargetMol
Ataluren is a novel, orally administered drug that targets nonsense mutations. Ataluren is approved for use by the European Medicines Agency to treat Duchenne Muscular Dystrophy in patients aged 5 years and older who are able to walk.
More Information
Supplier Page
Ataluren
100 mg
| Purity Not Available
TargetMol
Ataluren is a novel, orally administered drug that targets nonsense mutations. Ataluren is approved for use by the European Medicines Agency to treat Duchenne Muscular Dystrophy in patients aged 5 years and older who are able to walk.
More Information
Supplier Page